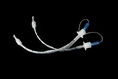
A reinforced uncuffed endotracheal tube is a sterile, flexible medical device crafted from high-quality silicone, integrated with a flexible wire spiral within its wall to resist compression and maintain airway patency. Designed without an inflatable cuff, it is specifically suited for patients who do not require airway sealing and rely on spontaneous breathing.
The silicone material ensures excellent biocompatibility, complying with ISO 10993 standards to minimize mucosal irritation, even during extended clinical use. Its smooth, rounded tip enables atraumatic insertion, reducing the risk of damage to delicate airway tissues. The embedded wire spiral provides robust structural support, preventing kinking or collapse even when the patient's head, neck, or body is repositioned-making it ideal for procedures or care scenarios that demand flexibility without compromising airflow. Available in multiple inner diameters (2.5-9.0mm) to accommodate pediatric and adult patients, the tube features an integrated radiopaque marker for precise placement verification via X-ray. Each unit undergoes rigorous quality testing, including compression resistance assessments, dimensional accuracy checks, and sterility validation, meeting global standards such as ISO 13485, CE, and NMPA certifications to ensure reliable performance in anesthesia, critical care, and surgical settings.
Product Specification
|
Item |
Description |
|
Applicable Population |
Pediatric and adult patients |
|
Insertion Route |
Oral/Nasal |
|
Cuff Type |
Uncuffed |
|
Core Structure |
Flexible wire spiral reinforced |
|
Material |
Sterile silicone (latex-free, flexible) |
|
Biocompatibility |
Complies with ISO 10993 |
|
Tube Inner Diameter |
2.5mm, 3.0mm, 3.5mm, 4.0mm, 4.5mm, 5.0mm, 5.5mm, 6.0mm, 7.0mm, 8.0mm, 9.0mm |
|
Radiopaque Marker |
Integrated (X-ray placement confirmation) |
|
Length |
Pediatric: 12-24cm; Adult: 22-34cm |
Product Properties

- Anti-compression & anti-kinking: Embedded wire spiral maintains airway patency against compression or bending.
- Cuff-free design: Suits patients requiring spontaneous breathing without airway sealing.
- Flexible silicone construction: Adapts to anatomical curves for patient comfort and unobstructed airflow.
- Airtraumatic insertion: Smooth tip minimizes irritation to airway mucosa.
- Sterile & biocompatible: Latex-free material reduces allergy risks; EO sterilized for immediate clinical use
Application
- Anesthesia: Airway support for procedures where spontaneous breathing is maintained (e.g., sedation, regional anesthesia).
- Critical care: Airway management for patients who do not require positive pressure ventilation or airway sealing.
- Pediatric care: Safe for children and infants with delicate airways, avoiding cuff-related mucosal compression.
- Surgical procedures: Ideal for surgeries involving head/neck repositioning (resists kinking during manipulation).
- Emergency care: Rapid airway access for patients needing temporary breathing support without sealed airways.

Physical Properties
|
Item |
Specification |
|
Compression Resistance |
Maintains patency under ≥20N external pressure |
|
Flexibility |
Sustains repeated bending (≥120°) without kinking/collapse |
|
Surface Smoothness |
Ra ≤0.6μm |
|
Weight |
Pediatric: 1-3g; Adult: 3-5g |
|
Sterility |
EO sterilized (3-year shelf life) |
QA&QC
- Certifications: Complies with ISO 13485, CE (EU MDR), and NMPA standards.
- Batch Testing: Each production batch undergoes compression resistance testing, dimensional accuracy verification, biocompatibility assessments (cytotoxicity, sensitization), and sterility validation.
- Traceability: Unique batch number and manufacturing date for full end-to-end quality tracking.
- Safety Documentation: Provides detailed Instruction for Use (IFU) and Material Safety Data Sheet (MSDS).

Packing,Storage,Handling and Transportation
The product is supplied in individual sterile ethylene oxide (EO) paper-plastic composite packaging for protection. Store in a clean, dry, and well-ventilated environment with a temperature range of -10℃~40℃ and relative humidity ≤80%, avoiding direct sunlight, high temperature, high humidity, and contact with corrosive substances or sharp objects. Only trained medical personnel are authorized to handle the product-prior to use, inspect the packaging for damage, tampering, or expiration; do not use if any defects are detected. During transportation, adhere to international and domestic medical device transportation regulations, ensuring protection against violent collisions, rain, and moisture to preserve product sterility and structural integrity.
Reasons for choosing us
Zhejiang Mediunion Healthcare Group Co. Ltd. (a branch of Evergrand Healthcare Group, founded in 2014) is located in Ningbo City, Zhejiang Province - enjoying an excellent location and convenient transportation. Our business covers medical device manufacturing & sales, plus investments in nursing homes, dental clinics, specialist hospitals and medical care services. Our products are sold worldwide, and we've won global consumer recognition with high-quality goods and comprehensive after-sales service.
Diversified Product Portfolio
Our product range includes disposable medical consumables, medical equipment and non-woven products (e.g. dental supplies, respiratory care items). This diverse lineup caters to the needs of medical institutions, clinics and other clients.

Reliable Supply & Collaboration
We have rich industry experience and partner with over 600 factories. These partners are equipped with advanced production equipment and skilled frontline staff, enabling us to produce customized products/components as per specific requests.

Strict Quality Assurance
We prioritize innovation & quality, backed by the ISO 13485:2016 quality management system. Strict control runs through raw material procurement, mass production, process management and finished product release. Our professional services (production control, order follow-up, delivery) also set us apart.
Certificate

FAQ
Q:Is your pricing EXW, FOB, CIF, or DDP?
A:We offer flexible pricing terms to meet your logistics needs, including EXW (Ex Works), FOB (Free On Board), CIF (Cost, Insurance, and Freight), and DDP (Delivered Duty Paid). You can choose the most suitable term based on your location, logistics preferences, and import capabilities, and our sales team will provide a detailed quotation accordingly.
Q:Can we order samples before bulk purchase?
A:Yes, we welcome sample orders before bulk purchase. We can provide 1-5 samples of standard products free of charge (you only need to cover the shipping cost). For customized samples, a small sample fee may apply, which will be deducted from your subsequent bulk order payment. Samples are typically shipped within 3-7 working days after confirmation.
Q:Are samples from mass production or lab batches?
A:All samples provided are from mass production batches, not lab batches. This ensures that the sample quality, performance, and specifications are fully consistent with the bulk products you will receive, allowing you to accurately evaluate the product's actual use effect and compliance.
Hot Tags: reinforced endotracheal tube uncuffed, China reinforced endotracheal tube uncuffed manufacturers, suppliers, factory, Adjustable Venturi Mask, DisposableNebulizer Mask, Nasal Cannula Soft Prong, Nasal Oxygen Catheter, Reinforced Endotracheal Tube Uncuffed, Soft Tracheostomy Mask